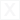
Working hours: Mon–Sat, 9:00 to 18:00. Orders accepted online: 24/7
Quantum Phytophos 25 ml
UAH 11.10
Quantum Phytophos 25 ml
Quantity
Full description
Quantum Fitofos Stop-Phytophthora - a highly concentrated microfertilizer with fungicidal properties for plant fertilization, disease prevention, and control, containing readily available forms of phosphorus and potassium for plant growth and development. The product induces a natural, sustained plant response against pathogens and stimulates the plant’s defense mechanisms and immune response against late blight, downy mildew, fusarium wilt, rhizoctonia, pythium, and others. The fertilizer is particularly effective for preventing these diseases. A special formulation of phosphorus and potassium with added organic acids allows plants to effectively utilize nutrients, stimulates root system development, and enhances the absorption of nutrients from the soil. “Quantum Phytophos” effectively combats diseases and serves as an accessible source of phosphorus and potassium.
Key properties of “Quantum-Fitofos”:
- The product contains a significant amount of phosphorus and potassium (25% P₂O₅ and 25% K₂O), which are available to plants both through the leaf surface and through the root system;
- It has a direct fungicidal effect on pathogens.
- The product induces a natural, long-lasting plant response against pathogens.
- The product’s components are easily absorbed by plants and transported via the phloem and xylem;
- The product inhibits the growth and development of pathogens, strengthens plant immunity, forms a defense mechanism and immune response, and provides additional opportunities for plant growth and development;
- The product contains slow-release forms of phosphorus and potassium for plant growth and development, ensuring increased yield, improved product quality, and enhanced fruit appearance.
Application method and dosage:
It is recommended to apply the product before the onset and development of diseases (as a preventive measure). In this case, the product is most effective.
All crops
Foliar application.
Fertilization and disease prevention:
Application rate per treatment: 2–2.5 L/ha.
Apply at 2–3 week intervals.
Do not apply at intervals of less than 3 days.
Fertilization and disease control:
Application rate per treatment: 4–5 L/ha.
Apply at 2–3 week intervals.
Do not apply at intervals of less than 3 days.
Working solution application rate: 250–300 L/ha,
(for vineyards and orchards: 500–1,000 L/ha).
Treatments are performed using standard sprayers.
It is not recommended to reduce the application rate of the working solution or to perform aerial spraying. Before mixing with other products, it is recommended to check their compatibility—a test preparation of the working solution should not produce a precipitate.
Slight cloudiness may occur.
Do not mix the product with copper-based fungicides or adhesives.
Do not use in cold (below +10°C) or hot (above +30°C) weather, or during intense sunlight.
All crops
Foliar application.
Fertilization and disease prevention:
Application rate per treatment: 2–2.5 L/ha.
Apply at 2–3 week intervals.
Do not apply at intervals of less than 3 days.
Fertilization and disease control:
Application rate per treatment: 4–5 L/ha.
Apply at 2–3 week intervals.
Do not apply at intervals of less than 3 days.
Working solution application rate: 250–300 L/ha,
(for vineyards and orchards: 500–1,000 L/ha).
Treatments are performed using standard sprayers.
It is not recommended to reduce the application rate of the working solution or to perform aerial spraying. Before mixing with other products, it is recommended to check their compatibility—a test preparation of the working solution should not produce a precipitate.
Slight cloudiness may occur.
Do not mix the product with copper-based fungicides or adhesives.
Do not use in cold (below +10°C) or hot (above +30°C) weather, or during intense sunlight.
The special phosphorus formulation “Quantum Phytophos” moves through the plant in both directions: it is absorbed by the roots and moves upward through the xylem, and is also absorbed by the leaves and transported downward through the phloem, whereas phosphorus in the form of phosphate can primarily move only through the xylem—from the roots upward to other parts of the plant.
Phosphites are a new formulation of phosphorus that provide phosphorus nutrition to plants while also offering fungicidal action.
Around the world, phosphite-based products have yielded excellent results both in terms of phosphorus nutrition and in the control and prevention of diseases.
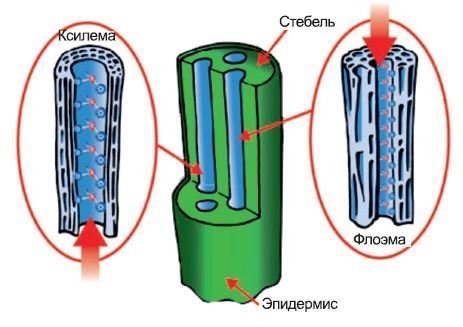
Scientists worldwide have recognized that phosphite-based products work effectively both as fertilizers and as fungicides. What, then, is the difference between phosphites (PO3) and traditional phosphates (PO4)?
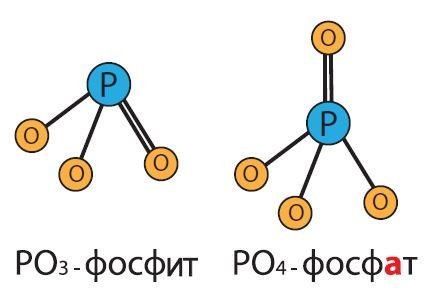
Phosphites are fundamentally different in nature from phosphates, which are the main component of granular fertilizers. Chemically, these compounds are similar, but phosphate contains one more oxygen atom, which significantly alters their properties and reactivity.
The effect of these two compounds on crops differs significantly, despite their chemical similarity. Phosphates form insoluble compounds in the soil, and only a small portion of the phosphorus is absorbed by plants. When applied to the leaf surface, phosphates are also poorly absorbed by the plant because they do not penetrate the cuticle layer of the leaf surface well.
Phosphite compounds (e.g., potassium phosphites) are usually used in liquid form—as an aqueous solution. Three oxygen atoms increase the mobility of phosphorus in plant tissues and in the soil. They can act on all parts of the plant—leaves, stem, and roots. Phosphites are easily absorbed and transported through the xylem and phloem to all parts of the plant.






































 Seeds
Seeds

















































